

Emerging research shows the impact of mitigation policies in schools. While it is too early to fully assess the direct impact of policies such as school mask mandates, several studies have started to give an idea of this impact.
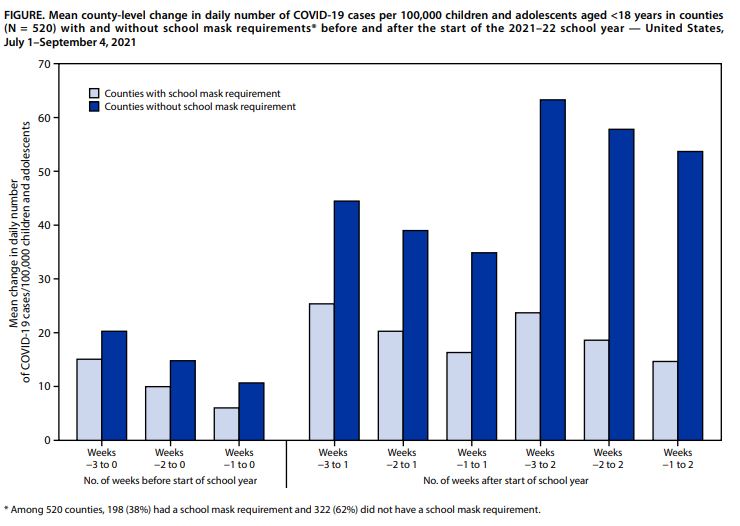
One recent study using CDC data assessed county case rates in schools with and without school mask requirements between July 1 and September 4, 2021. The study analyzed 520 counties across the U.S., and criteria for inclusion in the review requiring masks for all students and being open for at least 3 weeks. Results showed that there were larger increases in COVID-19 case rates among children in counties without school mask requirements compared to counties that had school mask requirements, even when researchers controlled for factors including age, race and ethnicity, vaccination rate, community transmission, population density, social vulnerability index score, COVID-19 community vulnerability index score, percentage uninsured, and percentage living in poverty.
Another study looked at the impact of mask policies on school-related outbreaks. In this study of 1,020 public schools in Arizona between July 15 and August 31, 2021, 21% of schools had established a mask requirement before school started (“early mask requirement”); 31% implemented a mask requirement after school started (“late mask requirement”); and 48% of schools had no mask requirement. Over the study period, there were 191 school-associated outbreaks: 8.4% of the outbreaks were in schools with early mask requirements; 32.5% in schools with late mask requirements; and 59% in schools without a mask requirement. Schools without mask requirements were 3.5 times more likely to have outbreaks compared to schools with mask requirements.
In May 2021, the Food and Drug Administration authorized the Pfizer COVID-19 vaccine for emergency use authorization in adolescents aged 12-15 (age 16+ was already approved under adult use). Since then, 350,881 North Carolina kids aged 12-17 have been vaccinated with at least one dose. The vaccine is available free of charge, regardless of insurance or immigration status.
On September 28, Pfizer and BioNTech announced that they had submitted data to the Food and Drug Administration indicating that their vaccine is safe and effective for children aged 5-11. The clinical trial, on more than 2,220 participants aged 5-11, showed safety and efficacy of a lower dose of the vaccine. The FDA has said that it will analyze this data as soon as possible, projecting that an authorization for the vaccine for children aged 5-11 may come within a few weeks.
However, some concern remains about how many parents will seek out the vaccine for their children. For adolescents aged 12-15 who are already eligible, only about 43% have been fully vaccinated nationwide, compared with 67% of adults. Among parents of children aged 5-11 surveyed by the Kaiser Family Foundation, 26% said they would vaccinate their children “right away” following FDA authorization, 40% said they would “wait and see,” and 25% said they would not vaccinate their child at all.
For vaccines for younger children, Pfizer has indicated that trial results for children aged 2-4 will be available before the end of 2021. They are also running a pediatric trial for children aged six months to 2 years old, with results expected a few months later. Moderna has also fully enrolled participants in a trial for children aged 6-11, and is conducting dose selection studies for younger age groups.
For children in North Carolina, risk mitigation policies such as masks, social distancing, and handwashing, especially within schools and child care settings, will continue to provide protection against COVID-19. Vaccines for adolescents and younger children will provide another effective tool to keep children and their families healthy and bring us closer to the end of this pandemic.
More NCIOM and NCMJ analysis on COVID-19, children, and families:
The American Rescue Plan Act: Provisions to Support Children and Families
Monitoring COVID-19 Impacts on Social Drivers of Health
2021 Child Health Report Card: Focus on the Impact of the COVID-19 Pandemic on NC Families
From Health Disparities to Hotspots: COVID-19 and the Drivers of Health